.png)
- Home
-
Industries
Industries We Serve
-
Medtech & Life Sciences
Driving innovation in healthcare and biotechnology through advanced medical devices and diagnostics.
-
Manufacturing & Industrial
Enabling smart manufacturing, automation, and operational efficiency at scale.
WHY US
Our product “Inventify” is a SaaS tool helping improving productivity and efficienc y in the supply chain. We help our customer cut through chaos in the business ecosystem. Starting from sales order automation from distributers, tracking each order from production to dispatch to the depots/ stores. We also enable our customers with channel finance maintaining the overall health of the supply chain.
-
-
Products
Products
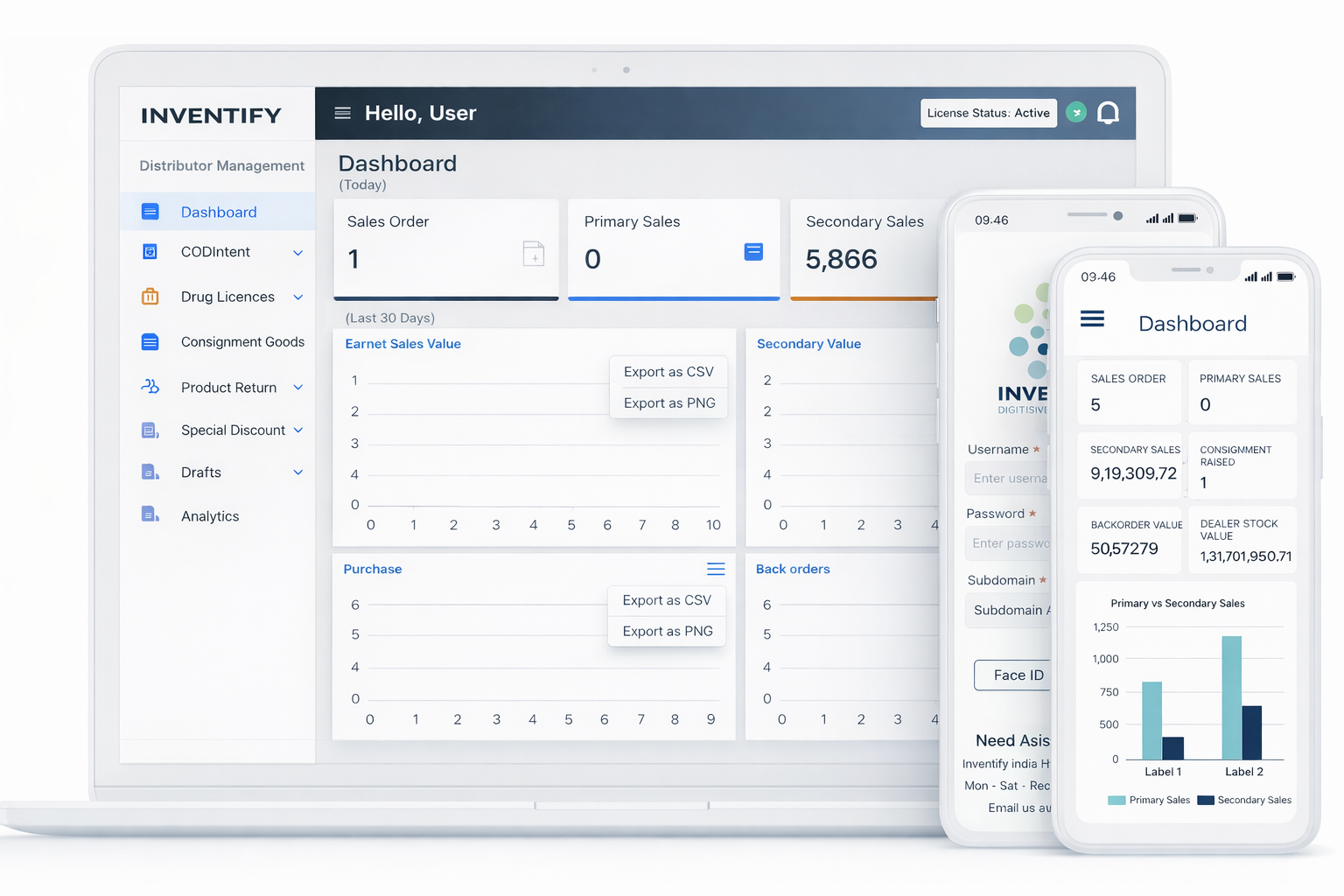
Distributor Management
Inventify DMS adoption presents tremendous opportunity to Medtech OEMs for growing revenues through traditional distribution channels, as more and more B2B businesses progressively move towards an omni-channel experience.
CRM Tool
Bring together every stage of customer and sales lifecycle — Lead → Account → Opportunity → Quotation → Deal Closure — into one streamlined, transparent system built for OEMs and its Distributors.
Account Planning
End-to-End Strategic & Operational Planning for Key and Major Accounts with a unified, dynamic account planning solution
Quotation Management
Transform the way you configure, price, approve, and track capital-equipment quotations.
Instrument Management
Track and manage instruments efficiently with complete lifecycle visibility.
Tender Management
From manual, fragmented tendering to a streamlined, compliant, and data-driven tender engine.
Service Management
End-to-end service & spare-parts visibility.
Revenue Assurance
A complete digital control stack for Revenue Assurance & Contract Intelligence for MedTech Channel Ecosystem
PLATFORM BENEFITS
- Improve channel visibility and drive smarter sales decisions.
- Reduce downtime and maximize asset performance.
- Enhance field efficiency and customer satisfaction.
- Speed up quoting and ensure pricing accuracy.
- Strengthen distributor collaboration and boost revenue.
- Increase win rates with streamlined tender processes.
- Turn market insights into actionable growth plans.
- Achieve complete service visibility and operational control.
- About Us
- Career
- Contact Us